Module 53 Randomization statistics
Learning goals
- Understand what randomization analyses are, and why they are awesome.
- Use randomization to determine if an outcome is significantly unexpected.
- Use randomization to compare two distributions.
- Use randomization to ask whether two variables are correlated
- Use randomization to determine the chances that a correlation is declining or increasing.
Basic idea
Classic significance statistics (i.e., p-values) are based on the frequentist framework in which you estimate the probability of an observed outcome based upon what would be expected if a null hypothesis were true. In a t-test, for example, you are asking, “What are the chances of observing what I have if these two sample sets were not meaningfully different?”
The key word in that first sentence is estimate. When frequentist theory was originally developed, statisticians did not have computers capable of generating vast amounts of random numbers. So they had to estimate p-values by developing strict rules and expectations about the way data are distributed. These rules had the result of (1) placing a lot of constraints on the way statistsics could be done, (2) forcing the development of increasingly specific tests for each novel sampling scenario, (3) confusing students trying to learn statistics, and (4) requiring the use of simplistic conventions, such as the 0.05 significance threshold, in order to make statistical results somewhat intelligible.
But in the age of R, null distributions no longer need to be theoretical, nor do p-values need to be hypothetical. We can actually calculate p-values based on real null distributions, thanks to the random number generators built into coding languages such as R. The practice of generating real null distributions based on your own specific dataset is known as randomization.
In a randomization routine, you shuffle the variables in your dataset to generate a distribution of outcomes that would be possible according to a null hypothesis. Most null hypotheses reduce to an expectation of no meaningful difference between two (or more) samples, which is to say that any differences between those samples are merely the product of random chance. Such randomly generated variability can be simulated easily in R.
Randomization may seem daunting at first, and it does indeed draw upon some of the higher-order skills in your R toolbag (such as custom functions, for loops, and sometimes even the apply() functions), but once you become oriented you will see how intuitive and simple these analyses can be.
The real advantages of randomization, however, are not for the programmer but for the reader. Randomization results are much easier to interpret and more information-rich than frequentist p-values. The advantages are many:
Rather than trusting a black box statistical test with obscure requirements about data distributions, you are performing the test yourself and building the test using your own data. You have more control and accountability, and no more risk of choosing the wrong statistical test. Because you are using your own data to produce the null distribution, you are able to retain the quirks of your own particular dataset; the limitations and inconsistencies in your sampling will be safely propagated into the statistic and properly reflected in the ucertainty of your results.
The interpretation of your p-value will be more intuitive. Rather than saying that some result is or is not significant based upon an arbitrary threshold such as 0.05, you can speak about the chances of meaningful results: “There is a 89% chance that these two sample sets are meaningfully different.” “There is a 92% chance that this trend is increasing.” These kinds of results are easier to interpret, and it avoids the need to make sense of p-values.
Finally, your statistical test returns much more than a p-value; it returns the null distribution too, which can be of enormous value in more complicated statistical analyses.
Generalized randomization workflow
Define your research question.
Define the null model that you wish to compare your observations against. This involves determining which variable you will need to shuffle.
Create a function that calculates the value of interest.
Use that function to determine the observed value of interest.
Use a
forloop to build a null distribution by shuffling your observations withR’s random number generators.Visualize your results to sanity-check what you are finding.
Calculate your p-value, by asking what proportion of the null distribution falls below the observed value.
The examples below will show you how to appy this generalized workflow. Note that we are purposefully applying randomization to scenarios in which classic statistical tests, such as t-tests and linear models, could also be used. We do so because those scenarios are relatively simple, they demonstrate the comparability of randomization to approaches you may already be familiar with, and they hint at the advantages of randomization over those other approaches. But the true value of randomization lies in its ability to carry out statistics when all other conventional tests fail; we hint at that value in the final use case.
Common use cases
1. Is outcome X significantly unexpected?
To practice this use case, let’s use the dataset humpback-whales.csv: 15 years of sightings of humpback whales within a fjord system in northern British Columbia, Canada (www.bcwhales.org).
Each individual whale in this dataset was identified using unique patterns on its tail, so each whale has an ID number.
nrow(hw)
[1] 4436
head(hw)
year sit bhvr id
1 2004 2004070701 BNF 42
2 2004 2004071401 BNF 42
3 2004 2004071401 BNF 59
4 2004 2004080301 BNF 30
5 2004 2004080401 MI-FE 106
6 2004 2004080401 MI-FE 114Each row shows a whale (column id) identified during a single sighting (column sit). Some sightings were of a single solo individual, but others were of groups of two or more individuals.
Our interest here is in these whales’ social behavior: do they have “friendships”, i.e., certain individuals that they prefer to associate with over others, or are they just randomly swimming around and bumping into one another?
Let’s tackle this one step at a time.
Step 1: Our research question is, Do any pairs of whales occur together an unexpectedly persistent social bonds? Since in science we are always limited by how well we sample the population of interest, a more precise version of our research question would be this: Based on this dataset, can we conclude whether or not any pairs of whales occur together an unexpectedly large number of times?
Step 2: The null model for this question is simple random mixing: there is no pattern to which whales occur in which group. So the variable that we will need to shuffle is the id column. With this approach, we be able to preserve the number of times each individual is seen and the total number of observations. So the limitations and inconsistencies in our sampling of each individual will be safely propagated into our statistics and properly reflected in the uncertainty of our results.
Step 3: So we need a function that returns the number of times two whales are associated. We will call this function association_tally().
association_tally <- function(hw,A,B){
siti <- hw$sit[hw$id==A]
sitj <- hw$sit[hw$id==B]
common_sits <- which(siti %in% sitj)
X <- length(common_sits)
return(X)
}Step 4: For our observed association tally, we will focus in on a single pair of humpback whales: IDs 59 and 11.
That’s alot of associations! Let’s see if that observation is statistically significant:
Step 5: The for loop for our null distribution will look like this:
nulls <- c()
for(i in 1:1000){
# save a safe copy of the data
hw_null <- hw
# shuffle the null variable
hw_null$id <- sample(hw_null$id,size=nrow(hw_null),replace=FALSE)
# determine null rate for this iteration
nulli <- association_tally(hw_null,A="59",B="11")
# save result
nulls[i] <- nulli
}Note! Here we only iterate our for loop 1,000 times, but most published randomizations are run for 5,000 to 10,000 iterations, just to ensure that the null distribution is adequately unbiased.
Step 6: To visualize these results, use a histogram:
par(mar=c(4.2,4.2,1,1))
hist(nulls,breaks=seq(0,25,by=1),xlim=c(0,(obs+10)),
main=NULL,xlab="Null distribution")
abline(v=obs,lwd=2,lty=3,col="blue")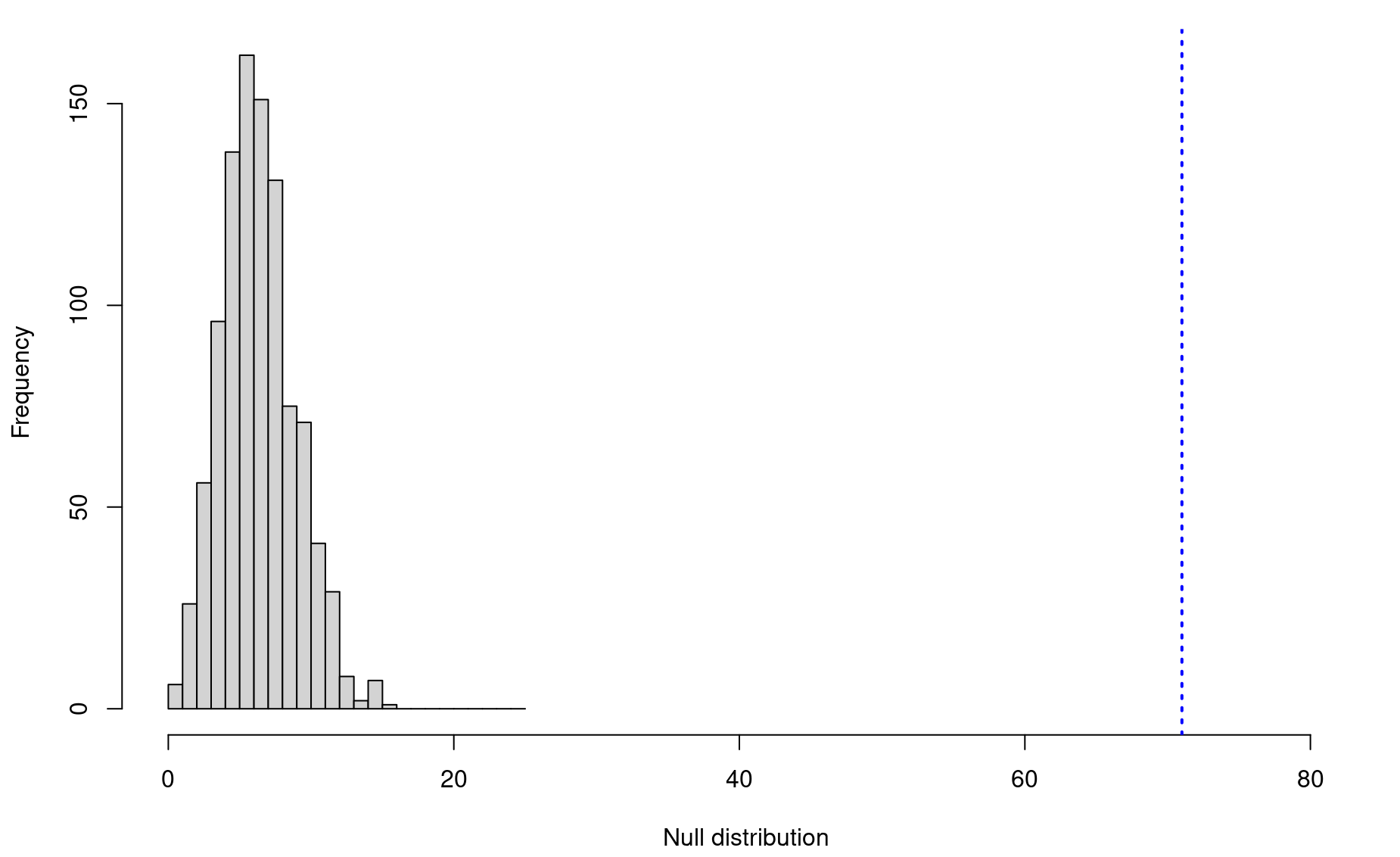
Step 7: Finally, we calculate our p-value:
Let’s do this again with a different humpback pair, IDs 16 and 118, who were seen together only 13 times. Is their association rate statistically significant?
# Individual IDs
A <- "58"
B <- "133"
# Observed association rate
obs <- association_tally(hw,A=A,B=B)
obs
[1] 9
# Generate null model
nulls <- c()
for(i in 1:1000){
hw_null <- hw
hw_null$id <- sample(hw_null$id,size=nrow(hw_null),replace=FALSE)
nulli <- association_tally(hw_null,A=A,B=B)
nulls[i] <- nulli
}
# Visualize
hist(nulls,breaks=seq(0,20,by=1),xlim=c(0,(obs+7)),
main=NULL,xlab="Null distribution")
abline(v=obs,lwd=2,lty=3,col="blue")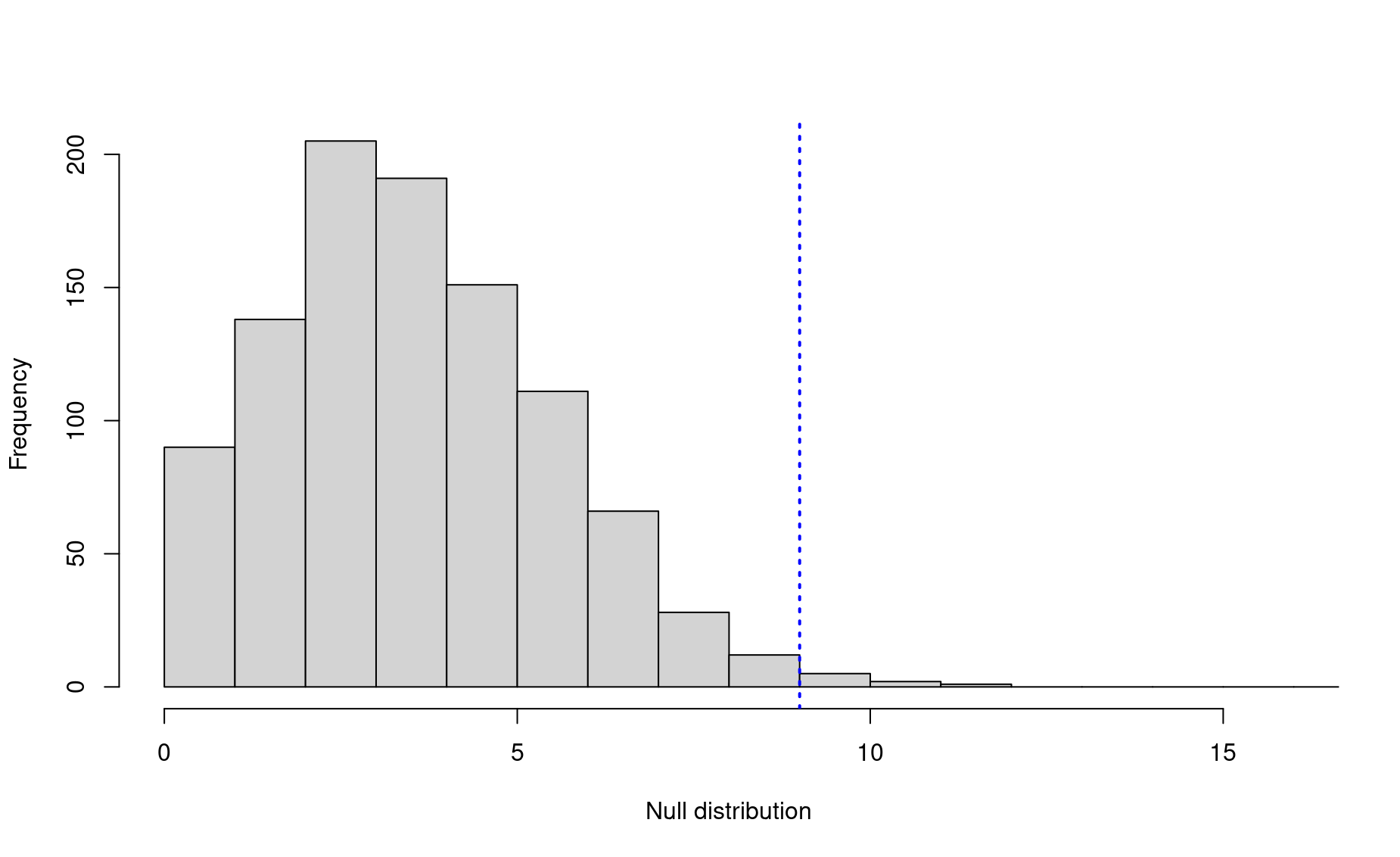
In this case, the observed association rate is still statistically significant at the conventional 0.05 level, but there is still an off chance that this is merely the result of random mixing.
2. Are two sample sets significantly different?
Building off of the whale scenario: Some of these whales have learned how to practice bubble-net feeding, a remarkable feeding strategy that requires group cooperation and special techniques for catching schooling fish such as herring. Other individuals have never been seen bubble net feeding. Are there any differences how often these two populations – the bubble netters and the “others” – use the study area?
First, let’s see how many of these individuals are known to bubble net feed:
# Establish the two populations
bnf_indices <- grep("BNF",hw$bhvr) # which detections are of BNFers
bnf_sits <- hw[bnf_indices,] # BNF sightings
bnf_ids <- unique(bnf_sits$id) # BNF individuals
length(bnf_ids)
[1] 108So, out of 132 individuals, 108 are known to practice bubble netting.
Let’s go through the steps:
1. Research question: Is there any difference in the number of years in which bubble-netters are seen compared to other whales?
2. Null model: In this case, the null model will be a distribution of average differences in the number of years in which individuals from the two populations are seen. This null distribution will be centered around zero. The variable we will shuffle is which individuals are known bubble netters, keeping the size of the two populations constant in each iteration.
3. Function:
annual_difference <- function(hw,bnf_ids){
bnf <- hw[hw$id %in% bnf_ids,] # Sightings of known BNFers
other <- hw[! hw$id %in% bnf_ids,] # Sightings of other whales
# Get distribution of years seen for individuals within each population
# sightings of each whale (rows) in each year (columns)
counts <- table(bnf$id,bnf$year)
# years seen for each individual
n_bnf <- apply(counts,1,function(x){length(which(x>0))})
# sightings of each whale (rows) in each year (columns)
counts <- table(other$id,other$year)
# years seen for each individual
n_other <- apply(counts,1,function(x){length(which(x>0))})
# Now get average difference between two distributions
mean_diff <- mean(n_bnf,na.rm=TRUE) - mean(n_other,na.rm=TRUE)
return(mean_diff)
}4. Observed value:
5. Build null distribution:
nulls <- c()
for(i in 1:1000){
bnf_null <- sample(unique(hw$id),size=length(bnf_ids),replace=FALSE)
nulli <- annual_difference(hw, bnf_ids=bnf_null)
nulls[i] <- nulli
}6. Visualize:
hist(nulls,breaks=seq(-5,5,by=.1),xlim=c(-5,5),
main=NULL,xlab="Null distribution")
abline(v=obs,lwd=2,lty=3,col="blue")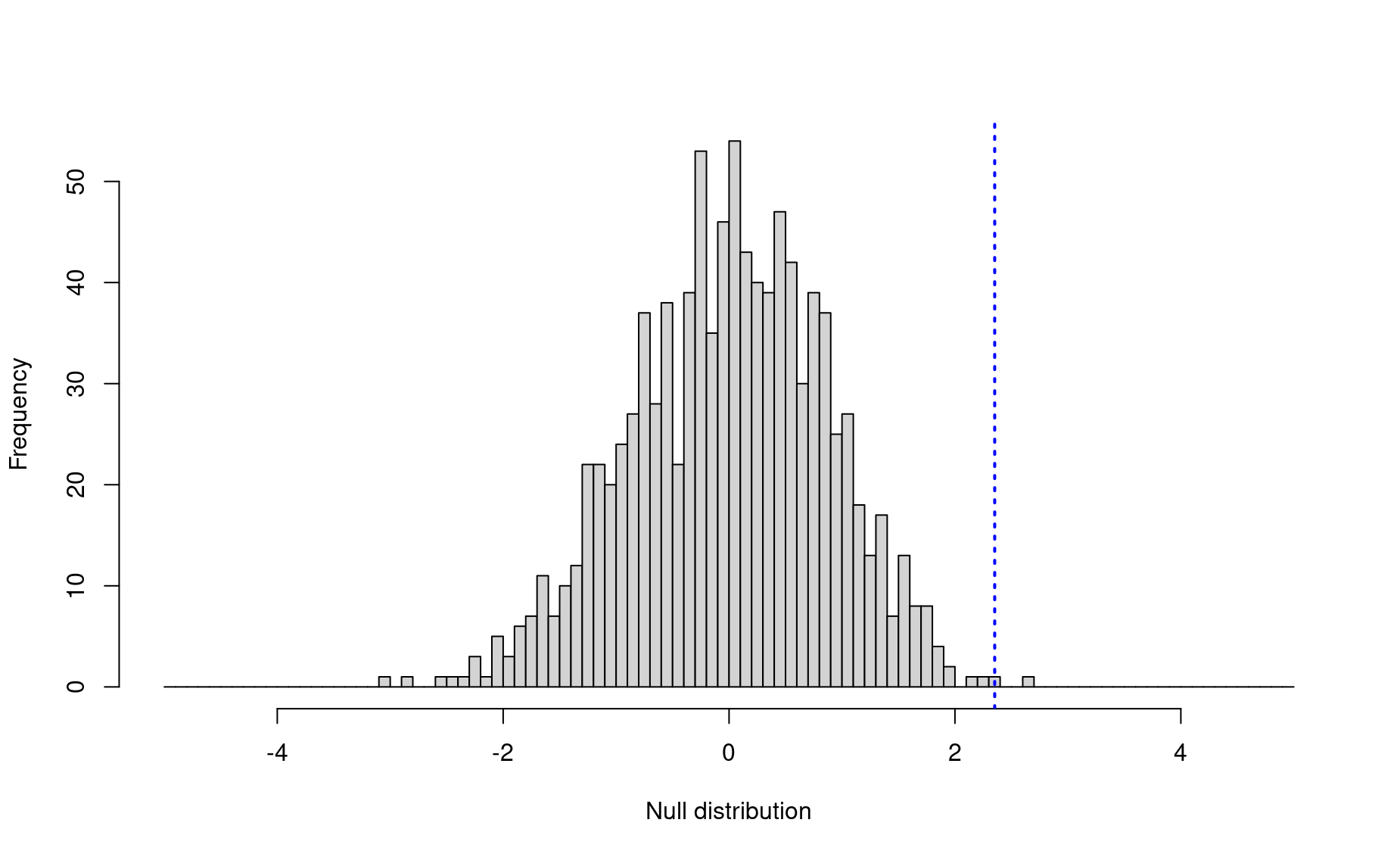
7. p-value:
So there is indeed a significant difference in the annual site fidelity in the two populations! Bubble net feeders practice higher annual return rates than other whales (p = 0.001).
4. Is the slope of a regression line negative?
Sometimes we have even less data about an endagered species. Let’s say we go out in 2021 for another porpoise survey and come up with these estimates from the species’ range:
library(truncnorm) ; set.seed(1234)
Error in library(truncnorm): there is no package called 'truncnorm'
n2021 <- rtruncnorm(n=25,a=0,mean=3.6,sd=3)
Error in rtruncnorm(n = 25, a = 0, mean = 3.6, sd = 3): could not find function "rtruncnorm"
n2021
Error in eval(expr, envir, enclos): object 'n2021' not foundWe want to know what the chances are that the population has declined between 2017 and 2021.
n2017 <- df$N[df$year==2017]
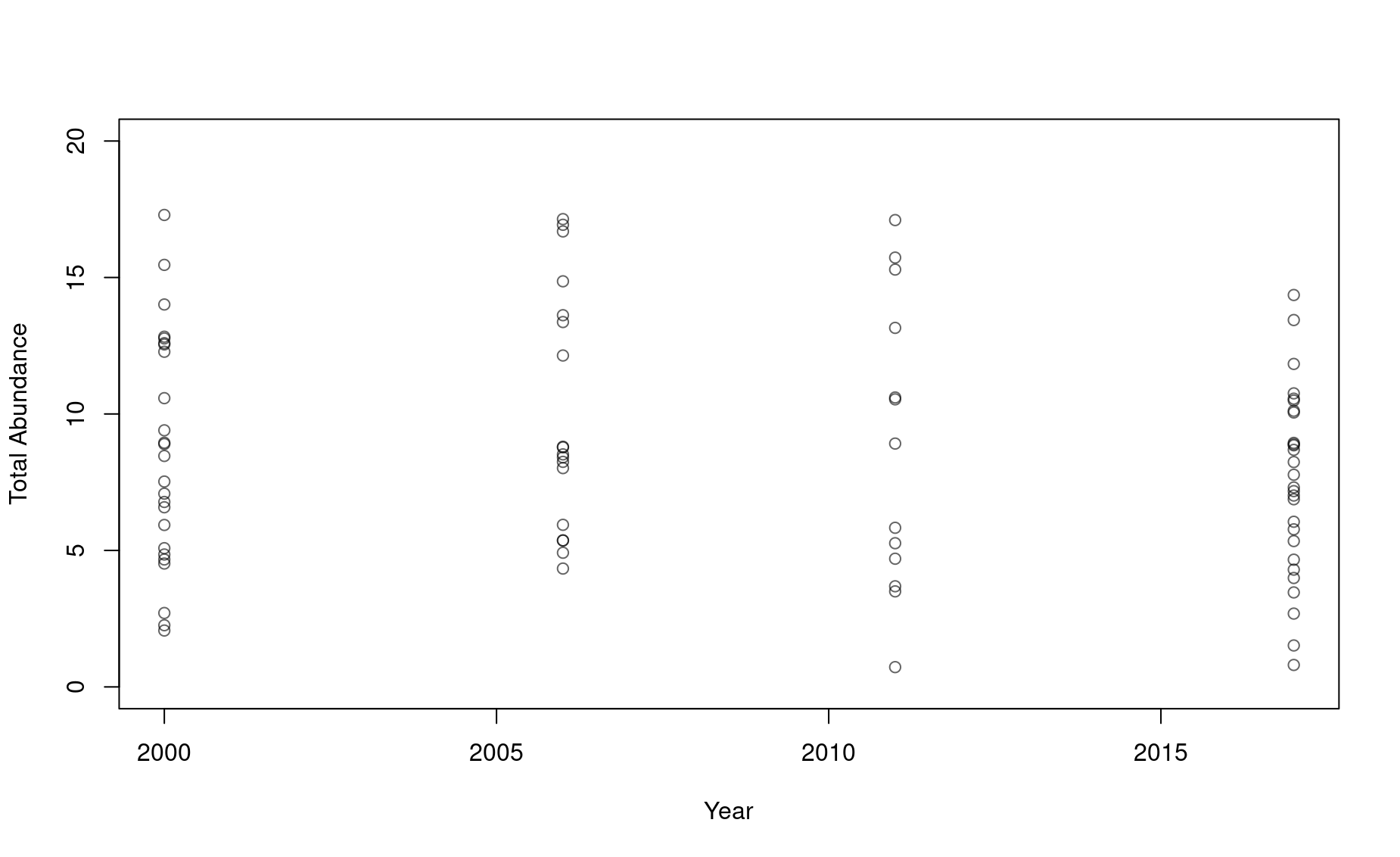
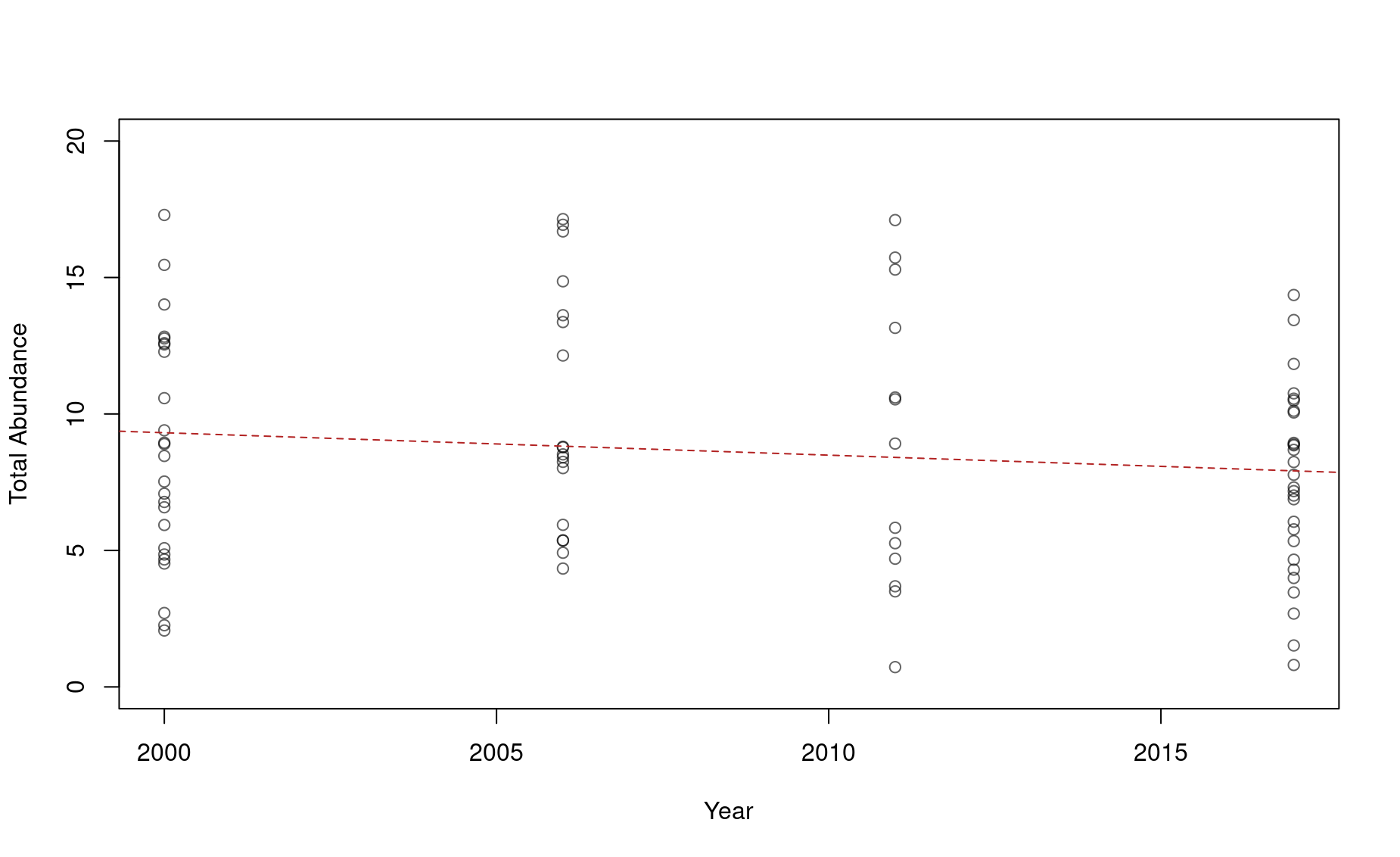
plot(1,type="n",ylim=c(0,15),xlim=c(2015,2023),
xlab="Year",
ylab="Total Abundance",
col=adjustcolor("black",alpha.f=.6))
points(n2017 ~ rep(2017,times=length(n2017)))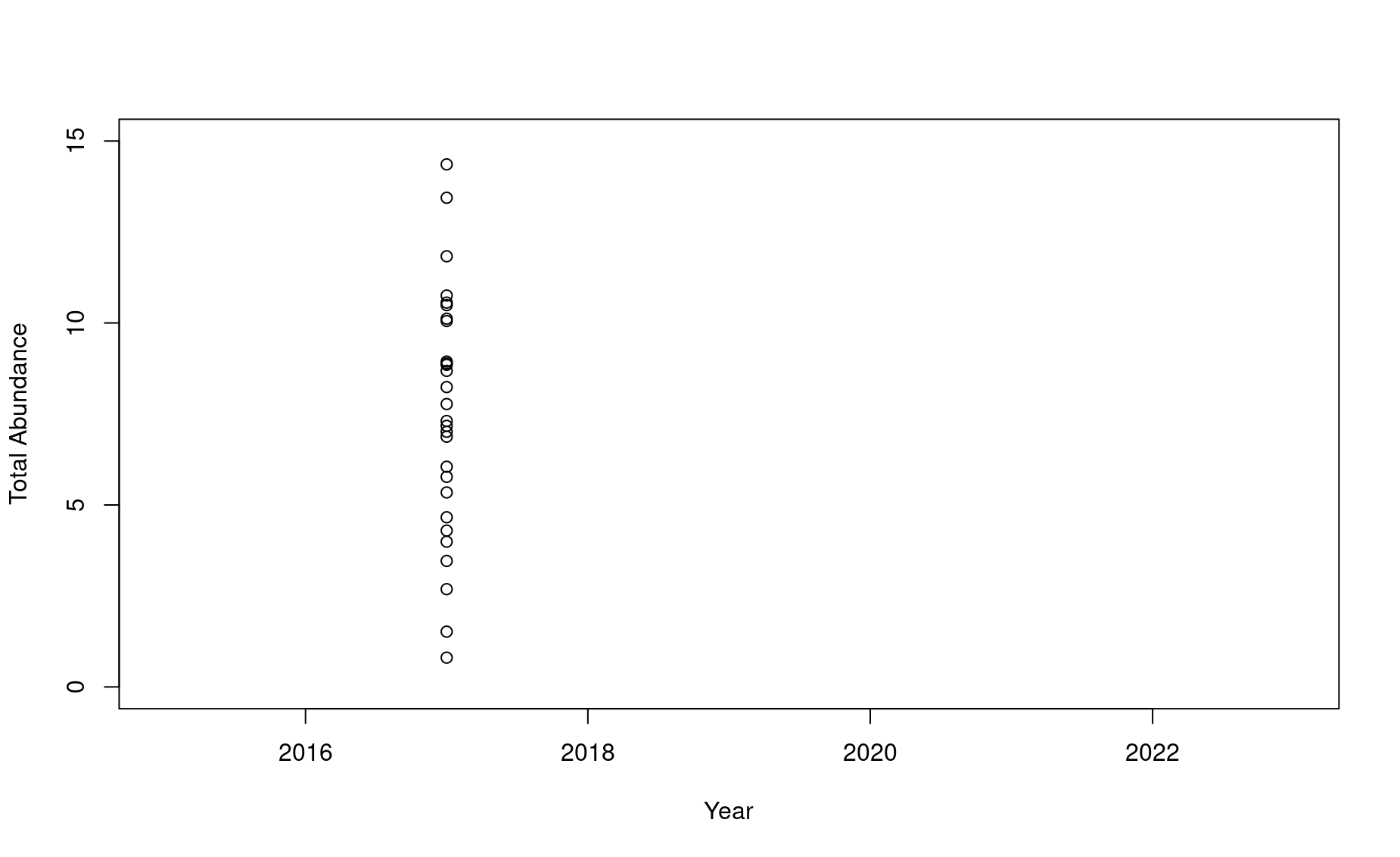
points(n2021 ~ rep(2021,times=length(n2021)))
Error in eval(predvars, data, env): object 'n2021' not foundIf this is all we have, what can we say about the status of this endangered species? With such a low sample size and so much variability, it can be really hard to produce significant or even compelling results. Well, what are the chances that this population is in decline? That is a question that a simple linear model cannot tell us.
In this case, our null model will be the distribution of differences of values drawn randomly from the two years’ datasets.
And in this randomization application, the workflow is a bit more simplified. We don’t need a core function, and we don’t even need a for loop.
# Randomly draw from the 2017 dataset 5,000 times
r2017 <- sample(n2017,size=10000,replace=TRUE)
# Do the same for the 2017 dataset
r2021 <- sample(n2021,size=10000,replace=TRUE)
Error in sample(n2021, size = 10000, replace = TRUE): object 'n2021' not found
# Get the difference of these two datasets
diffs <- r2021 - r2017
Error in eval(expr, envir, enclos): object 'r2021' not found
# Visualize
hist(diffs,breaks=seq(-20,20,by=1),xlim=c(-20,20),
main=NULL,xlab="Null distribution",border="grey70")
Error in hist.default(diffs, breaks = seq(-20, 20, by = 1), xlim = c(-20, : some 'x' not counted; maybe 'breaks' do not span range of 'x'
abline(v=0,lwd=2,lty=3,col="blue")
Error in int_abline(a = a, b = b, h = h, v = v, untf = untf, ...): plot.new has not been called yet
# Get p_value
p_value <- length(which(diffs >= 0)) / length(diffs)
# Convert to chances of decline
chances_of_decline <- 100*(1 - p_value)
chances_of_decline
[1] 8.333333The chances that this population is in decline is 8.3333333%.